| A | B |
|---|
What type of electromagnetic radiation is in the "A" spot?,  | gamma rays,  |
What type of electromagnetic radiation is in the "B" spot?,  | X-rays,  |
What type of electromagnetic radiation is in the "C" spot?,  | UV rays,  |
What type of electromagnetic radiation is in the "D" spot?,  | visible light,  |
What type of electromagnetic radiation is in the "E" spot?,  | infrared,  |
What type of electromagnetic radiation is in the "F" spot?,  | microwaves,  |
What type of electromagnetic radiation is in the "G" spot?,  | radiowaves,  |
Visible light has a wavelength range between ______ at the violet end and _____ at the red end.,  | 380 nanometers (violet end), 750 nm at the red end,  |
| Violet light has ____ energy, _____ wavelength and ______ frequency compared to red light. | violet light has greater energy, shorter wavelength and higher frequency compared to red light |
| The frequency of electromagnetic radiation is inversely proportional to ______ but directly proportional to _____. | frequency is inversely proportional to wavelength but directly proportional to the energy of the ray. |
| When electromagnetic energy hits matter, it can either be _____, _____, or _____ depending on the wavelength of energy and type of matter. | absorbed, reflected, or transmitted |
| When electromagnetic energy hits matter, it can either be absorbed, reflected, or transmitted depending on the _______ and _____. | wavelength of energy and the type of matter |
The red arrow signifies ______., 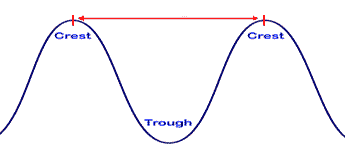 | The wavelength of this wave,  |
| Name the 7 colors of from longest wavelengths to shortest wavelengths. | red, orange, yellow, green, blue, indigo, violet (Remeber ROY-G-BIV) |
| Name the 7 colors of from shortest wavelengths to longest wavelengths. | Violet, Indigo, Blue, Green, Yellow, Orange, Red (Remember that the violet end of the visible light spectrum has the most energy (think UV radiation) and therefore the shortest wavelength) |
| Which type of electromagnetic radiation has the highest energy? | gamma rays |
| Which type of electromagnetic radiation has the highest frequency? | gamma rays |
| Which type of electromagnetic radiation has the longest wavelength? | radiowaves |
| Which type of electromagnetic radiation has the shortest wavelength? | gamma rays |
| Which type of electromagnetic radiation has the lowest frequency? | radiowaves |
| Which type of electromagnetic radiation has the lowest energy? | radiowaves |
| The type of electromagnetic radiation that you would find having a wavelength slightly shorter than violet light would be _______. | ultra vioet (UV) radiation |
| The type of electromagnetic radiation that you would find having a wavelength slightly longer than red light would be _______. | infrared |
| Infrared radiation has a slightly ______ wavelength than red light. | longer |
| Ultra violet (UV) radiation has a slightly ______ wavelength than violet light. | shorter |
| The frequency of electromagnetic waves is _______ proportional to the energy of those waves. | directly |
| The energy of electromagnetic waves is _______ proportional to the frequency of those waves. | directly |
| The wavelength of electromagnetic waves is _______ proportional to the frequency of those waves. | inversely |
| The frequency of electromagnetic waves is _______ proportional to the wavelength of those waves. | inversely |
| he wavelength of electromagnetic waves is _______ proportional to the energy of those waves. | inversely |
| The energy of electromagnetic waves is _______ proportional to the wavelength of those waves. | inversely |
| The higher the frequency of waves, the ______ the energy of those waves. | higher |
| The longer the wavelength of a wave, the _____ the frequency of those waves. | lower |
| The longer the wavelength of a wave, the _____ the energy of those waves. | lower |
| The lowest energy state of an atom is called its _______ state. | ground |
| An atom that absorbs energy moves from the ground state to the ______ state. | excited |
| Electromagnetic radiation can be thought of as little packets of energy called _______. | photons |
| A _______ of electromagnetic energy has a specific wavelength. | photon |
The picture to the right shows an ______________., 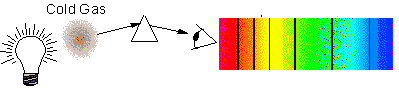 | absorption-line spectrum, 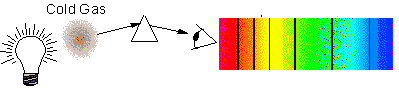 |
The picture to the right shows an ______________., 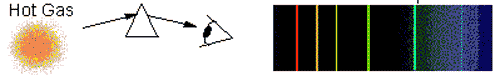 | emission-line spectrum, 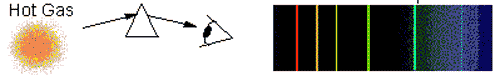 |
| What is the equation that relates the frequency of a wave to its energy? | E = hv (where E = energy, h = Planck's constant, and v= frequency) |
| What is the equation that relates wavelength, frequency and the speed of light? | wavelength X energy = the speed of light |