| A | B |
|---|
| _______ forces are the forces of attraction between molecules. | Intermolecular |
| Intermolecular forces are the forces of attraction _________ molecules. | between |
| To change from a liquid to a gas, molecules must be able to escape the _________ that attract each molecule to its neighboring molecules in the liquid. | intermolecular forces |
| Molecules with greater intermolecular forces between them have _____ melting and boiling points. | higher |
| Covalent bonds that hold atoms together within a molecule are called ______ forces. | intramolecular ("intra" means "within" while "inter" means "between.") |
| The intermolecular forces attract one molecule to another molecule are generally ____ than the intramolecular forces that attract atoms to each other within the same molecule. | weaker |
| Covalently bonded substances generally have _____ melting and boiling points than ionically bonded substances because _______ between molecules are generallly weaker than the ionic bonds that hold ionic substances together. | lower, the intermolecular forces (when molecules melt or boil, the covalent bonds that hold a molecule don't break like the ionic bonds of ionic substances. Instead, the weaker intermolecular bonds break, allowing molecules to move relative to their neighboring molecules) |
| The strongest intermolecular forces exist between ______ molecules. | polar |
| What are the three general types of intermolecular forces? List the strongest type first and the weakest type last. | 1) Dipole-dipole 2) Dipole-induced dipole 3) induced dipole - induced dipole (a.k.a. London Dispersion Forces) |
| Another name for intermolecular forces is _________. | Van der Waals forces |
| Another name for Van der Waals forces is ______. | intermolecular forces |
| Another name for induced dipole - induced dipole forces is ______. | London Dispersion forces. |
| Another name London Dispersion forces is ______. | induced dipole - induced dipole forces. |
| What is the strongest type of intermolecular force? | dipole - dipole forces |
| Dipole - dipole forces exist between a _____ molecule and a _____ molecule. | polar, polar |
| Dipole - Induced Dipole forces exist between a _____ molecule and a _____ molecule. | polar, non-polar |
| Induced Dipole - Induced Dipole forces exist between a _____ molecule and a _____ molecule. | non-polar, non-polar |
| The weakest type of intermolecular force is a(n) ______. | induced dipole -induced dipole force (a.k.a. - London Dispersion Force) |
| Polar molecules are said to have a ______ which is created by equal but opposite charges in different ends of the molecule. | dipole |
| A particularly strong type of dipole - dipole force that is caused by the attraction of hydrogen bonded to either oxygen, nitrogen or fluorine in one molecule, and either oxygen, fluorine, or nitrogen atoms in another molecule. | hydrogen bonds (the pink dotted lines in the picture below represent hydrogen bonds that hold complementary nucleotides together in DNA),  |
What are three words to describe the bonds between the water molecules (represented by the black dotted lines) in the picture below? List them in order from least specific to most specific.,  | Intermolecular (or Van der Waals) forces, dipole - dipole force, hydrogen bond,  |
| The tendency of certain liquids (like water) to move up a narrow tube due to the attraction of the liquid molecules to the surface of the tube is called _________. | capillary action, 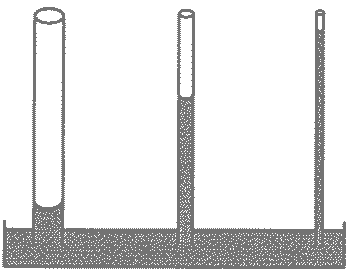 |
Water tends to bead up do to ______ of the molecules within the bead. This term means "sticking to self.",  | cohesion,  |
| A polar molecule can _____ a dipole in a nonpolar molecule by temporarily attracting its electrons to one side of the non-polar molecule. | induce |
When molecules stick to the side of a surface (like the water molecules in the side of the menscus below sticking to the glass in the graduated cylinder), they are exhibiting the property of ______.,  | adhesion (the water molecules are adhering to the glass, causing the meniscus to rise up slightly on the sides),  |
The water strider in the picture below doesn't break through the surface of the water because of the strong ________ caused by hydrogen bonding of water molecules at the surface.,  | surface tension,  |
| Water is best at dissolving ____ and ____ substances. | polar, ionic |
| Water (a polar molecule) is able to dissolve oxygen gas (a non-polar molecule) because of which type of intermolecular force between the two? | dipole - induced dipole |