| A | B |
|---|
According to the chart below, visible light has a wavelength range between ______ at the violet end and _____ at the red end.,  | 380 nanometers (violet end), 750 nm at the red end,  |
Violet light has ____ energy, _____ wavelength and ______ frequency compared to red light.,  | violet light has greater energy, shorter wavelength and higher frequency compared to red light,  |
The frequency of electromagnetic radiation is inversely proportional to ______ but directly proportional to _____.,  | frequency is inversely proportional to wavelength but directly proportional to the energy of the ray.,  |
| When electromagnetic energy hits matter, it can either be _____, _____, or _____ depending on the wavelength of energy and type of matter. | absorbed, reflected, or transmitted |
| When electromagnetic energy hits matter, it can either be absorbed, reflected, or transmitted depending on the _______ and _____. | wavelength of energy and the type of matter |
The red arrow signifies ______., 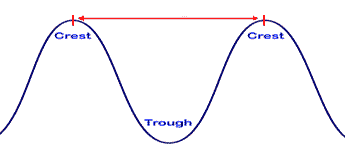 | The wavelength of this wave,  |
Which type of electromagnetic radiation has the highest energy?,  | gamma rays,  |
Which type of electromagnetic radiation has the highest frequency?,  | gamma rays,  |
Which type of electromagnetic radiation has the longest wavelength?,  | radiowaves,  |
Which type of electromagnetic radiation has the shortest wavelength?,  | gamma rays,  |
Which type of electromagnetic radiation has the lowest frequency?,  | radiowaves,  |
Which type of electromagnetic radiation has the lowest energy?,  | radiowaves,  |
The type of electromagnetic radiation that you would find having a wavelength slightly shorter than violet light would be _______.,  | ultra violet (UV) radiation,  |
The type of electromagnetic radiation that you would find having a wavelength slightly longer than red light would be _______.,  | infrared,  |
Infrared radiation has a slightly ______ wavelength than red light.,  | longer,  |
Ultra violet (UV) radiation has a slightly ______ wavelength than violet light.,  | shorter,  |
The frequency of electromagnetic waves is _______ proportional to the energy of those waves.,  | directly,  |
The energy of electromagnetic waves is _______ proportional to the frequency of those waves.,  | directly,  |
The wavelength of electromagnetic waves is _______ proportional to the frequency of those waves.,  | inversely,  |
The frequency of electromagnetic waves is _______ proportional to the wavelength of those waves.,  | inversely,  |
he wavelength of electromagnetic waves is _______ proportional to the energy of those waves.,  | inversely,  |
The energy of electromagnetic waves is _______ proportional to the wavelength of those waves.,  | inversely,  |
The higher the frequency of waves, the ______ the energy of those waves.,  | higher,  |
The longer the wavelength of a wave, the _____ the frequency of those waves.,  | lower,  |
The longer the wavelength of a wave, the _____ the energy of those waves.,  | lower,  |
| The lowest energy state of an atom is called its _______ state. | ground |
| An atom that absorbs energy moves from the ground state to the ______ state. | excited |
| Electromagnetic radiation can be thought of as little packets of energy called _______. | photons |
| A _______ of electromagnetic energy has a specific wavelength. | photon |
| The transformation of a substance, or substances into one or more new substances is known as a _______. | chemical reaction |
| Which law states that "mass is neither created nor destroyed during ordinary chemical reactions or physical changes." | The law of conservation of mass |
| If the mass of the reactants in a chemical reaction is 3.11 grams, what will the mass of the products be after the chemical reaction? | 3.11 grams (The same as the mass of the reactants, as predicted by the law of conservation of mass) |
| Mass is neither _____ nor ______ during ordinary chemical reactions or physical changes. | created, destroyed |
| The fact that a chemical compound contains the same elements in exactly the same proportions, regardless of the size of the sample or source of the compound is known as the ______. | law of definite proportions |
| If two or more different compounds are composed of the same two elements (for example, CO and CO2) then the ratio of the masses of the second element combined with a certain mass of the first of the first element is always a ratio of small whole numbers. This statement is known as ______. | The law of multiple proportions (For example, if 1.00 grams of carbon is used to make carbon monoxide, it will react with 1.33 grams. If 1.00 grams of carbon is used to make carbon dioxide, then it will react with 2.66 grams of oxygen. The ratio of oxygen used in carbon dioxide to oxygen used in carbon monoxide is 2.66 to 1.33, or 2 to 1, thus the ratio of small whole numbers) |
| Who disproved Dalton's theory that atoms are the smallest possible particles? | Chuck Norris when he smashed one with his fist. |
| A(n) _____ is the smallest particle of an element that retains the chemical properties of that element. | atom |
| The ____ is a very small region located in the center of the atom. | nucleus |
| The nucleus is made up of at least one positively charged particle called a(n) _____ and usually one or more neutral particles called _____. | proton, neutron |
| Surrounding the nucleus is a region occupied by negatively charged particles called _____. | electrons |
| Protons, neutrons, and electrons are often referred to as ______ particles. | subatomic |
| Thomson's ________ experiments measured the charge-to-mass ratio of an electron. | cathode-ray tube,  |
| ________ was able to determine the charge-to-mass ratio of an electron with his cathode-ray tube experiments. | J.J. Thomson,  |
| _________ was able to measure the charge of an electron with his oil drop experiments. | Robert Millikan |
| Millikan was able to measure the charge of an electron with his ______ experiments. | oil drop |
| Rutherford's _____ foil experiments led to the discovery of the nucleus. | gold foil,  |
| _______ discovered the nucleus with his gold foil experiment (1911). | Ernest Rutherford,  |
| Most of the alpha particles that Rutherford shot at the thin gold foil ______, indicating that atoms are mostly empty space. | passed through,  |
| Most of the alpha particles that Rutherford shot at the thin gold foil passed through, indicating that atoms are mostly _______. | empty space,  |
| The fact that a small percentage of the alpha particles that Rutherford shot at the thin gold foil seemed to bounce back was evidence for the presence of ______ in atoms. | a small dense nucleus,  |
| The fact that a small percentage of the alpha particles that Rutherford shot at the thin gold foil seemed ________ back was evidence for the presence of a small dense nucleus in atoms. | bounce back,  |
| Except for the simplest type of hydrogen atom, all atomic nuclei are made of _________. | protons and neutrons (the simplest type of hydrogen atom has one proton and no neutrons) |
| The positive charge of a(n) _______ is _______ in magnitude to the negative charge of a(n) ________. | proton, equal, electron |
| A neutron has a ____ charge. | neutral |
| Each element has a different number of _____ in their nucleus. | protons |
| The short-range forces that hold the particles of a nucleus together when they are very close to one another are called _____. | nuclear forces |
| The atomic ______ is the distance from the center of the atom to the outer portion of the electron cloud. | radius |
| Atomic radii are usually measured in _______. | picometers |
What is "A" pointing to and what is its charge?,  | an electron, negative,  |
What is "B" pointing to and what is its charge?,  | The nucleus (positive because it consists of positively charged protons and neutral neutrons),  |
What is "C" pointing to and what is its charge?,  | proton, positive,  |
What is "D" pointing to and what is its charge?,  | neutron, neutral,  |
| What is smaller than an electron? | Chuck Norris' sense of fear (but no one has been brave enough to discover it yet) |
| Atoms of different elements have different numbers of __________. | protons |
| Atoms of the same element alwaysl have the________ number of protons | same |
| The ______ of an element is the number of protons in each atom of that element | atomic number |
| ________ are atoms of the same element that have different masses. | Isotopes |
| The isotopes of a particular element all have the same number of ________ and ________ but different numbers of __________. | protons, electrons, neutrons |
| The mass number is the total number of _______ and _________ that make up the __________ of an isotope | protons and neutrons in the nucleus |
| The number of protons and neutrons in the nucleus of an atom is equal to the atom's _______. | mass number |
| How would you write the hyphen notation of an isotope of carbon that had a mass number of 14? | carbon-14 |
| ______ is the general term for a specific isotope of an element. | Nuclide |
| If you are given the mass number and the atomic number of an element, how do you find the number of neutrons in the nucleus? | # of neutrons = mass# - atomic# |
| If you are given the mass number and the atomic number of an element, how do you find the number of neutrons in the nucleus? | # of protons = the atomic number |
| If you are given the number of protons and the number of neutrons in an element, how do find the mass number? | add the # of protons to the # of neutrons |
| If you are given the number of protons and the number of neutrons in an element, how do find the atomic number? | The atomic number = the number of protons |
| If you are given the number of protons and the number of neutrons in an element, how do find the name of the element? | Look on the periodic table for the element with an atomic number that = the number of protons you were given |
| If you are given the mass number and the atomic number of an element, how do you find the name of that element? | Look on the periodic table for the element with the same atomic number. The mass number is irrelevent. |
The number 19 on the periodic table box below is the ____ of potassium.,  | atomic number,  |
The number 39.0938 is the _____ of potassium.,  | average atomic mass |
According to the image below, how many protons does potassium have?,  | 19 (you can tell by the atomic number),  |
How many electrons would an atom of potassium have?,  | 19 (the same as the number of protons because atoms are electrically neutral),  |
| How would you write the nuclear symbol for an isotope of Lithium that has 3 protons and 4 neutrons in its nucleus? | .,  |
The graphic below shows the nuclear symbol for an isotope of lithium. What does the 7 and the 3 refer to?,  | The 7 is the mass number and the 3 is the atomic number,  |
How many protons and how many neutrons are in this isotope of lithium?,  | 3 protons and 4 neutrons,  |
| The average atomic mass of an element is the weighted average of all naturally occuring _______ of that element. | isotopes |
| What is the equation for calculating average atomic mass? | Average atomic mass = atomic mass of isotope 1 X percent composition of isotope 1 + atomic mass of isotope 2 X percent composition of isotope 2 + atomic mass of any other isotopes X % composition of those isotopes, etc. |
| How many particles are in 1 mole of a substance? | 6.022 X 10 to the 23rd power |
| How do you determine the molar mass of an element? | Find the average atomic mass of that element on the periodic table and make the unit grams per mole. |
| What is Avogadro's number expressed to 4 significant digits? | 6.022 X 10 to the 23rd power |
| What is the abbreviation for moles? | mol |