| A | B |
|---|
| The first person to name the basic particle an "atom" was the ancient Greek philosopher ______. | Democritus |
| Which law states that "mass is neither created nor destroyed during ordinary chemical reactions or physical changes." | The law of conservation of mass |
| If the mass of the reactants in a chemical reaction is 3.11 grams, what will the mass of the products be after the chemical reaction? | 3.11 grams (The same as the mass of the reactants, as predicted by the law of conservation of mass) |
| Mass is neither _____ nor ______ during ordinary chemical reactions or physical changes. | created, destroyed |
| Who disproved Dalton's theory that atoms are the smallest possible particles? | Chuck Norris when he smashed one with his spinning round-house kick. If you are the first one to email me saying "Chuck Norris Rules!!!" I will give you an extra credit point on the next quiz which will be called the Atomic Theory Quiz.,  |
| Thomson's ________ experiments helped him discover the presence and charge of electrons | cathode-ray tube,  |
________ discovered electrons.,  | J.J. Thomson,  |
| Ernest Rutherford's _________ experiments led to the discovery of the nucleus. | gold foil,  |
_______ discovered the nucleus with his gold foil experiment (1911).,  | Rutherford,  |
Most of the alpha particles that Rutherford shot at the thin gold foil ______, indicating that atoms are mostly empty space.,  | passed through,  |
Most of the alpha particles that Rutherford shot at the thin gold foil passed through, indicating that atoms are mostly _______.,  | empty space,  |
The fact that a small percentage of the alpha particles that Rutherford shot at the thin gold foil seemed to bounce back was evidence for the presence of ______ in atoms.,  | a small dense nucleus,  |
The fact that a small percentage of the alpha particles that Rutherford shot at the thin gold foil seemed to ________ was evidence for the presence of a small dense nucleus in atoms.,  | bounce back,  |
| Who created the first periodic table in 1869? | Mendeleev |
| The first periodic table was based on the ordering of elements by increasing _______. | atomic mass (today, it goes by increasing atomic number) |
| The first periodic table had several empty spaces because ______. | the elements that fit in those empty spaces hadn't been discovered yet (they were within the next 15 years) |
| Who was the first person to organize the periodic table according to increasing atomic number? | Moseley (in 1911); Mendeleev had organized his periodic table by increasing atomic mass |
| Thomson proposed the ________ model of the atom in which he envisioned positive and negative charged particles of the atom to be spread out evenly. | plum pudding, 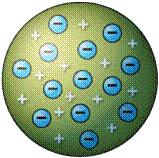 |
____________ were shot at gold foil, most of which passed through.,  | Alpha particles,  |
| The ________ model of the atom postulated that electrons travel in fixed orbits around the nucleus. | Bohr |
| Electrons with more potential energy are _________ the nucleus than electrons with less potential energy. | further away |
| Electrons that are closer to the nucleus have _____ potential energy than electrons that are further away from the nucleus. | less |
| Electrons must _______ energy to move to orbits that are further away from the nucleus | absorb |
| Electrons _______ energy when they move to lower energy levels. | emit (same as "lose" or "give off" energy) |
| When electrons fall back down to energy levels that are closer to the nucleus, energy is released in a process called _____. | emission |
| What was still wrong about Dalton's atomic theory? | He thought atoms were the smallest possible particle and couldn't be further divided (we now know that atoms are made of sub-atomic particles like protons, neutrons and electrons) |
| Who stated that when atoms combine, they do so in simple whole-number ratios? | Dalton (It was part of his atomic theory) |
| How did Mosely improve the organization of the periodic table? | He arranged it according to increasing atomic # (the number of protons in the nucleus) instead of increasing average atomic mass like Mendeleev did with his original periodic table) |