| A | B |
|---|
| Acid-Base Indicators | A weak acid or a weak base. The undissociated form of the indicator is a different color than the iogenic form of the indicator.,  |
| End Point | The conclusion of a chemical reaction, especially during a titration in which no more titrant should be added (can be indicated by a color change or appearance of a precipitate).,  |
| Equivalence Point | Also called stoichiometric point, of a chemical reaction occurs during a chemical titration when the amount of titrant added is equivalent, or equal, to the amount of analyte.,  |
| pH | The logarithm of the reciprocal of hydrogen-ion concentration in gram atoms per liter; provides a measure on a scale from 0 to 14 of the acidity or alkalinity of a solution ( 7 is neutral, than 7 is more basic, less than 7 is more acidic), 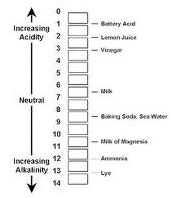 |
| pOH | Measures the basicity of a solution. It is the negative log of the concentration of the hydroxide ions.,  |
| Primary Standard | A chemical that can be obtained in an unvarying state and used as an approved example against which other examples of the same chemical can be compared.,  |
| Self-Ionization of Water | Is the chemical reaction in which two water molecules react to produce a hydronium ion (H3O+) and a hydroxide ion (OH-):,  |
| Standard Solution | Any solution which has a precisely known concentration. Similarly, a solution of known concentration has been standardized.,  |
| Titration | The process, operation, or method of determining the concentration of a substance in solution by adding to it a standard reagent of known concentration in carefully measured amounts until a reaction of definite and known proportion is complete, 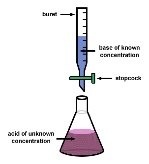 |
| Transition Interval | In a titrimetric analysis, the range in concentration of the species being determined over which a variation in a chemical indicator can be observed visually., 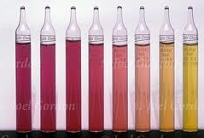 |
| pH Meter | Is an electronic instrument used to measure the pH (acidity or alkalinity) of a liquid, 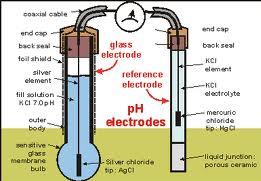 |