| A | B |
|---|
| Amphoteric | having the characteristics of an acid and a base and capable of reacting chemically either as an acid or a base.,  |
| Neutralization | A reaction between acid and base which neutralizes both and results in the formation of water plus a salt, 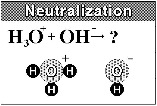 |
| Bronsted Lowry Base | any substance that can donate a hydrogen ion (proton), 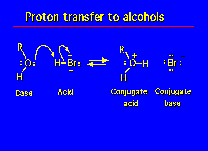 |
| Lewis Base | any substance capable of forming a covalent bond with an acid by transferring a pair of electrons to it., 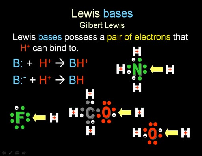 |
| Lewis Acid- Base Reaction | molecular entity (and the corresponding chemical species) that is an electron-pair acceptor and therefore able to react with a Lewis base to form a Lewis adduct, by sharing the electron pair furnished by the Lewis base., 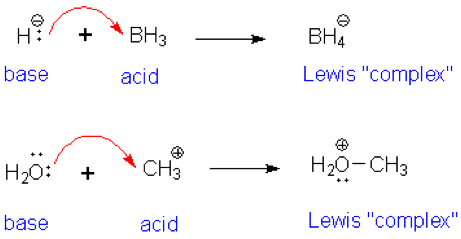 |
| Binary Acid | A binary compound in which H is bonded to one or more of the more electronegative nonmetals,  |
| Oxyacid | An acid containing oxygen, as chloric acid or sulphuric acid; contrasted with the hydracids, which contain no oxygen, as hydrochloric acid.,  |
| Arrhenius Acid | A substance that dissociates in aqueous solution to produce hydrogen ions, 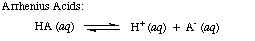 |
| Arrhenius Base | A substance that dissociates in aqueous solution to produce hydroxide ions, 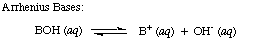 |
| Strong Acid | an acid that is completely dissociated in an aqueous solution., 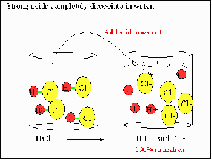 |
| Weak Acid | an acid that is partially dissociated in an aqueous solution., 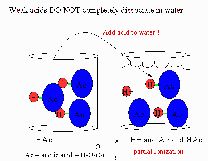 |
| Bronsted-Lowry Acid | a material that gives up hydrogen ions during a chemical reaction.,  |
| Bronsted-Lowry Acid-Base Reaction | a material that accepts hydrogen ions during a chemical reaction., 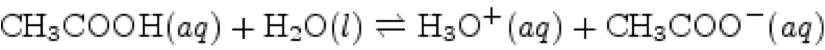 |
| Monoprotic Acid | Acid that can form only one hydronium ion per molecule; may be strong or weak. Acid that contains one ionizable hydrogen atom per formula unit., 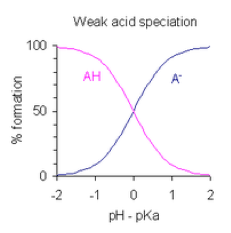 |
| Polyprotic Acid | an acid that can give up more than one hyrogoen atom. H 2 SO 4 for example, 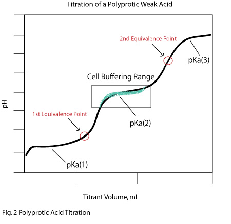 |
| Diprotic Acid | acid such as H2SO4 (sulfuric acid) that happens to contain within its molecular structure two hydrogen atoms per molecule capable of dissociating in water,  |
| Triprotic Acid | An acid that has three ionizable hydrogen atoms in each molecule, 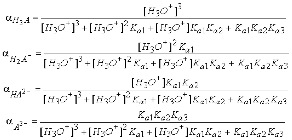 |
| Lewis Base | A compound which can act as an electron pair donor,  |
| Conjugate Base | The structure that results when a Bronsted-Lowry acid donates a proton,  |
| Conjugate Acid | The structure that results when a Bronsted-Lowry base accepts a proton, 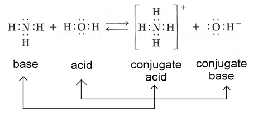 |
| Salt | ionic compounds which can result from the neutralization reaction of acids,  |