| A | B |
|---|
| Name the first 10 alkanes. | methane, ethane, propane, butane, pentane, hexane, heptane, octane, nonane, decane |
| Straight chain hydrocarbons with double bonds are called ______. How would you name one with 3 carbons? | alkenes, propene (Technically, if there are 2 double bonds, it would be propadiene, but don't worry about that for now) |
| Straight chain hydrocarbons with triple bonds are called _____. How would you name one with 2 carbons? | alkynes, ethyne |
What is the name of this molecule?, 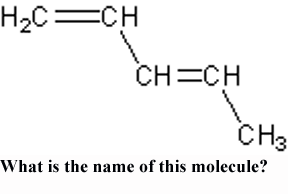 | 1,3-Pentadiene (This name indicates with the "1,3" that the double bonds come off the 1st and 3rd carbons and there are two double bonds "di." We know they are double bonds because of the "-ene" ending),  |
What is the name of this molecule?, 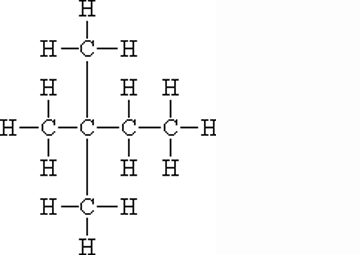 | 2,2-dimethyl-butane (Can also be called iso-hexane because of its six carbons, but this name doesn't give you enough information to figure out the structure. The "2,2 dimethyl" part of the name tells us that there are two methyl groups coming off the 2nd carbon in a butane molecule),  |
What is the name of this molecule?,  | 2,3-dimethyl-butane Can also be called iso-hexane because of its six carbons, but this name doesn't give you enough information to figure out the structure. The "2,3 dimethyl" part of the name tells us that there is a methyl group coming of the second carbon in a butane molecule as well as the third),  |
What type of molecule is this?, 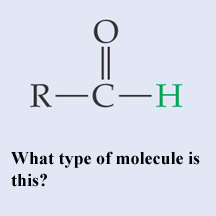 | A basic aldehyde pp64&65,  |
What type of molecule is this?,  | carboxylic-acid pp64&65,  |
What type of molecule is this?, -copy.gif) | aldehyde (hexanal) pp64&65, -copy.gif) |
What type of molecule is this?, .gif) | ketone (2-butanone) pp64&65, .gif) |
What type of molecule is this?, 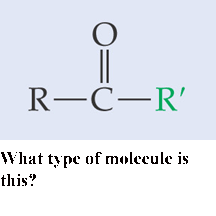 | Ketone pp64&65, 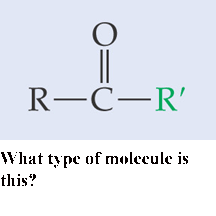 |
| C6H6 in a ring structure is _____. | Benzene p61, 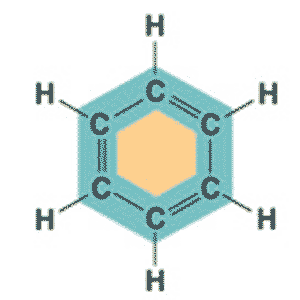 |
| What is the molecular formula and shape of benzene? | C6H6, ring p61 |
| One of several organic compounds with the same molecular formula but different structures, and therefore, different properties. | isomer p62 |
| A functional group present in organic acids consisting of a single carbon atom double-bonded to an oxygen atom and also bonded to a hydroxyl group. | carboxyl group pp64&65,  |
| A functional group consisting of a sulfur atom bonded to a hydrogen atom (--SH). | sulfhydryl group pp64&65,  |
| A functional group that is often written as --COOH. | carboxyl group (It's -COOH because it is a C double-bonded to an O and single-bonded to OH) pp64&65,  |
| A functional group that consists of a nitrogen atom bonded to two hydrogen atoms. It can act as a base in a solution, accepting a hydrogen ion and acquiring a charge of +1. | amino group pp64&65, 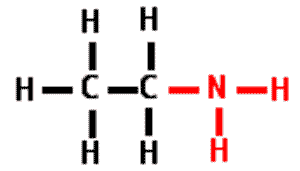 |
| An organic molecule consisting only of carbon and hydrogen is called a(n) _____. | hydrocarbon (the butane molecule below is a hydrocarbon because it has only hydrogens and carbons) p61, 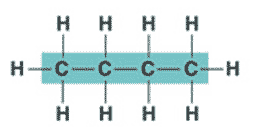 |
| An arrangement of two non-carbon atoms, each bound to one of the carbons in a carbon-carbon double bond,where the two non-carbon atoms are on the same side relative to the double bond. | cis p62, 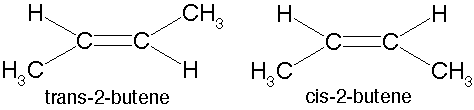 |
| An arrangement of two non-carbon atoms, each bound to one of the carbons in a carbon-carbon double bond,where the two non carbon atoms are opposite sides relative to the double bond. | trans p62, 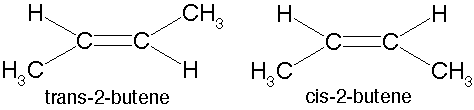 |
| A specific configuration of atoms commonly attached to the carbon skeleton of organic molecules and usually involved in chemical reactions. | functional group (the carboxyl-group in red below is a functional group that makes the molecule acidic because it often loses the hydrogen as a hydrogen ion into the solution which lowers the pH of the solution) p63,  |
| A type of isomer in which carbons have covalent bonds to the same atoms, but these atoms differ in their spatial arrangements due to the inflexibility of carbon-carbon double bonds. | Cis-trans isomers (a.k.a. geometric isomer) p62, 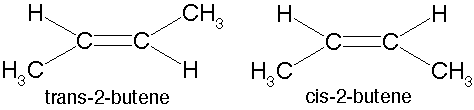 |
| A functional group that is important in energy transfer. | Phosphate group (think of ATP-->ADP + P + energy) p66, 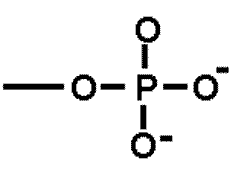 |
| One of several organic compounds that have the same molecular formula but differ in the covalent arrangement of their atoms. | structural isomer p62, 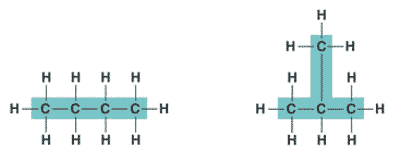 |
| A functional group present in aldehydes and ketones that consists of a carbon atom double-bonded to an oxygen atom. | carbonyl group pp64&65, 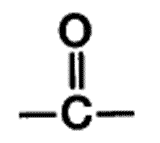 |
| A functional group consisting of a hydrogen atom joined to an oxygen atom by a polar covalent bond. Molecules possessing this group are soluble in water and are called alcohols. | hydroxyl group pp64&65,  |
| One of two molecules that are mirror images of each other. | enantiomer (also called a stereo-isomer) p62, 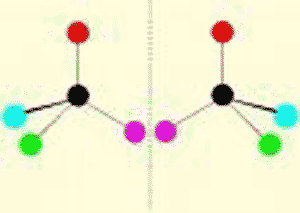 |
| A(n) ______ carbon is a carbon atom that is attached to four different atoms or groups of atoms. The result is to allow for the possibility of another molecules that can be an enantiomer (mirror image) of the first molecule. | asymmetric carbon (In the diagram below, the #2 carbon is the asymmetric carbon. Even though it bonds to two carbons, the #1 carbon can be considered an aldehyde GROUP and the number #3 carbon can be considered a propyl group. Therefore, all 4 groups coming off the #2 carbon are different) p62, 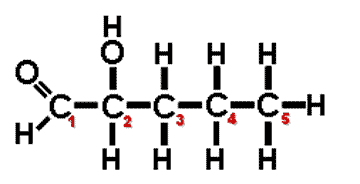 |
What is the name of the molecule shown below?, 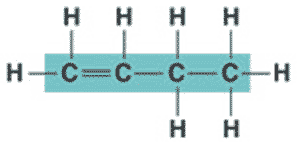 | 1-butene (the 1 signifies that the double bond starts at the first carbon),  |
What is the name of the molecule shown below?, 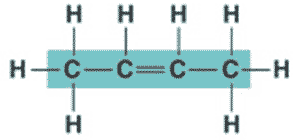 | 2-butene (the 2 signifies that the double bond starts at the second carbon),  |
What type of molecule is this?,  | An aldehyde (It's an aldehyde because the carbonyl group is coming off a terminal carbon. If it wasn't, it would be a ketone. The name of this molecule is ethanal, since it has two carbons like ethane) pp64&65,  |
What is the name of the functional group in red?,  | amino group pp64&65,  |
Which carbon on this molecule is the asymmetrical carbon?,  | In the diagram below, the #2 carbon is the asymmetric carbon. Even though it bonds to two carbons, the #1 carbon can be considered an aldehyde GROUP and the number #3 carbon can be considered a propyl group. Therefore, all 4 groups coming off the #2 carbon are different. p62,  |
What is the name of this ring structure?, 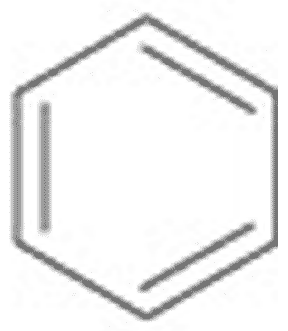 | Benzene (The structure you saw on the other side of this card was an abbreviated structural formula. These formulas assume a carbon at each corner and enough hydrogens to make 4 bonds coming off each carbon. Below is the unabbreviated structural formula for comparison) p63,  |
What is the name of this ring structure?,  | Benzene p63,  |
What is the name of this hydrocarbon?,  | Butane,  |
What is the name of this functional group?,  | carbonyl group pp64&65,  |
What is the name of this type of molecule and what is the name of its functional group?, 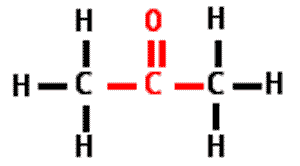 | ketone, carbonyl group (The specific name of the molecule pictured is propanone because it is a ketone with 3 carbons like propane) pp64&65,  |
What is the name of this type of molecule and what is the name of its functional group?,  | Carboxylic acid, carboxyl group (This molecule is called ethanoic acid but is more commonly referred to as acetic acid which is the acid in vineger) pp64&65,  |
What is this diagram showing?, 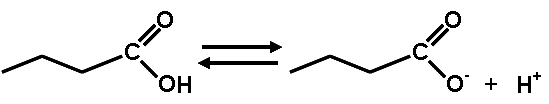 | This shows how carboxylic acids (notice the carboxyl group at the end of the abbreviated structural formula of hydrocarbons) will release a hydrogen ion into water solutions. Hydrogen ions decrease the pH of the solution. That is why it is a carboxylic ACID. pp64&65, 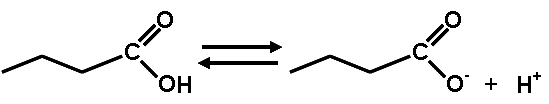 |
What is the name of this molecule?,  | cyclohexane (The structure you saw on the other side of this card was an abbreviated structural formula. These formulas assume a carbon at each corner and enough hydrogens to make 4 bonds coming off each carbon. Below is the unabbreviated structural formula for comparison) p61, 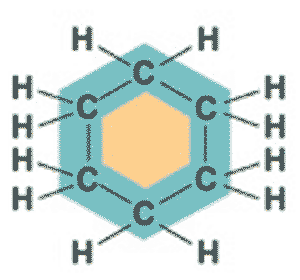 |
What is the name of this molecule?,  | cyclohexane p61,  |
What do we call these two molecules?,  | enantiomers (or stereoisomers; notice that they are mirror images of each other) p62,  |
What type of isomers are these?, 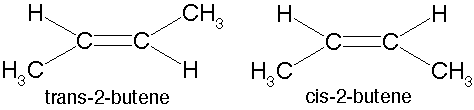 | cis-trans isomers (a.k.a. geometric isomers) p62, 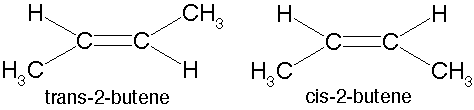 |
Which of these cis-trans isomers would be considered the "cis" isomer and which would be the "trans" isomer?,  | The one on the left is "cis" and the one on the right is "trans" (the prefix trans means across) p62,  |
What is the name of this hydrocarbon?,  | Ethane,  |
What is the name of this type of molecule and what is the name of its functional group?,  | An alcohol, hydroxyl group (This is ethanol because it has two carbons like ethane. The common name is ethyl alcohol) p64&65,  |
What is the name of this functional group?,  | phosphate group p64&65,  |
What is the name of this hydrocarbon?, 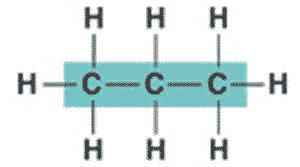 | propane,  |
What type of isomers are these?,  | structural isomers p62,  |
What is the name of this functional group?,  | Sulfhydryl group p64&65,  |
What were the results and conclusions of Stanley Millers 1953 experiment (pictured below)?,  | Stanley Miller tried to recreate the conditions of early earth by placing some simple compounds like water, hydrogen, methane and ammonia in a sealed container. He added energy in the form of electricity to simulate lightning and found that these primitive Earth gasses reacted and formed a variety of organic compounds that would have been necessary for the first life to form. p59,  |
| Carbon can form a wide variety of compounds because it forms ____ covalent bonds. | 4 (p60) |
Each line in a structural formula (like the one below) represents a _____., 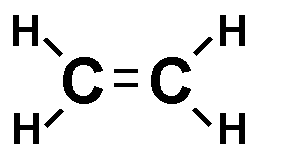 | pair of shared electrons, 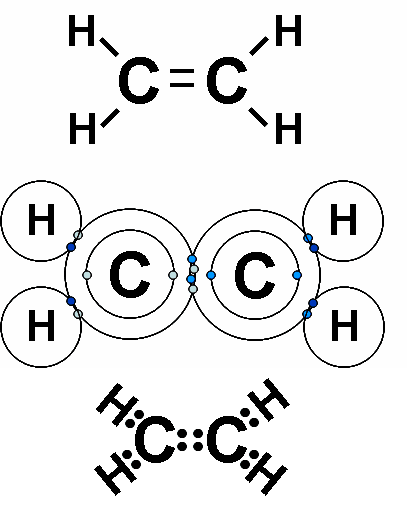 |
| ____ is the source of carbon for all of the organic molecules found in organisms. | carbon dioxide p58 |
Hydrocarbons (like the one shown below) are ______ and therefore don't dissolve in water.,  | Non-polar or hydrophobic p61 |
| An important energy storing molecule that has 3 phosphate groups is called _____. | Adenosine tri-phosphate (ATP) p66, 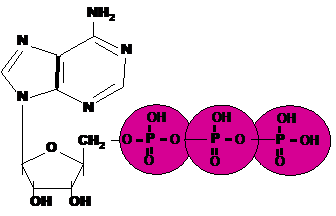 |
| Hydrocarbons can be either _____, _____, or _____ shaped. | straight, branched, or ring shaped p61 |
| Organic molecules consisting of only carbon and hydrogen are called ______. | hydrocarbons p61 |
., 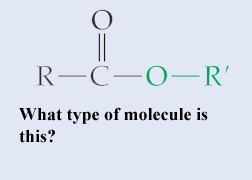 | Ester {The image below is an ester called propyl-ethanoate (you don't need to know how to name these specifically, but you might be able to look at the prefixes involved and figure out the logic). Esters are known for their aromatic qualities (smell) and are used in many perfumes, pheromones, and help give flavor and smell to certain fruits, like apples, bananas, and strawberries. When you take college organic chemistry, you will probably make one of these fruit-smelling ester molecules by mixing an organic acid and an alcohol. Your book doesn't cover this, but I have seen references in AP tests to phosphodiester bonds. These are bonds where a phosphate group helps link one nucleotide to another in DNA or RNA. Knowing what an ester bond is can help you figure out what a phosphodiester bond is}, .gif) |
What type of bond is shown being formed in the picture below?, 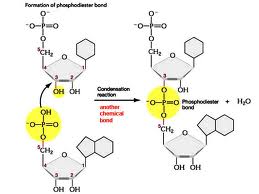 | phosphodiester bond (These are bonds involving a phosphate group (PO4-) that link one nucleotide to another in DNA or RNA. Notice the part of the bond that gives it the ester name. This is not covered in your book, but I have seen it on AP exams, so learn it),  |