| A | B |
|---|
| temperature | measure of the average kinetic energy of the particles of an object |
| Celsius temperature scale | scale in which water freezes at 0 degress and boils at 100 degrees,  |
| Kelvin temperature scale | temperature scale in which the lowest reading is 0, the lowest temperature possible (absolute zero),  |
| absolute zero | lowest possible temperature at which all molecular motion stops |
| calorie | amount of heat needed to raise the temperature of 1 gram of water 1 degree Celsius |
| joule | unit of energy, including heat energy, in the metric system |
| Specific heat | the specific heat of a material is the amount of heat energy required to raise the temperature of 1 gram of the material, one degree Celsius; the symbol (c) denotes specific heat an equation |
| Specific heat of water | 1 calorie or 4.18 (4.2) joules/g * degrees C |
| Kelvin to Celsius | Kelvin = 273 + degrees Celsius |
| Calorimetry | the study of energy changes involved in chemical reactions |
| activation energy | the amount of energy required to initiate a reaction |
| calorimeter | an apparatus used to measure the heat change in a chemcal reaction, 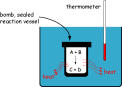 |