| A | B |
|---|
| * Name the first 10 alkanes. | methane, ethane, propane, butane, pentane, hexane, heptane, octane, nonane, decane (from an activity that will completed in class) |
| * Straight chain hydrocarbons with double bonds are called ______. How would you name one with 3 carbons? | alkenes, propene (Technically, if there are 2 double bonds, it would be propadiene, but don't worry about that for now) |
| * Straight chain hydrocarbons with triple bonds are called _____. How would you name one with 2 carbons? | alkynes, ethyne |
** What is the name of this molecule?, 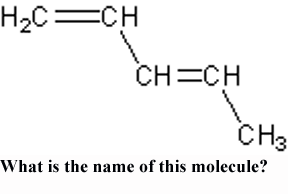 | 1,3-Pentadiene (This name indicates with the "1,3" that the double bonds come off the 1st and 3rd carbons and there are two double bonds "di." We know they are double bonds because of the "-ene" ending),  |
** What is the name of this molecule?, 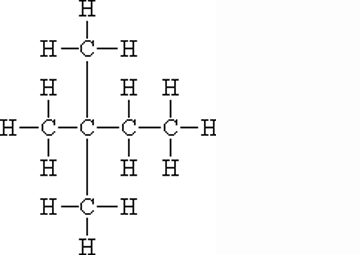 | 2,2-dimethyl-butane (Can also be called iso-hexane because of its six carbons, but this name doesn't give you enough information to figure out the structure. The "2,2 dimethyl" part of the name tells us that there are two methyl groups coming off the 2nd carbon in a butane molecule),  |
** What is the name of this molecule?,  | 2,3-dimethyl-butane Can also be called iso-hexane because of its six carbons, but this name doesn't give you enough information to figure out the structure. The "2,3 dimethyl" part of the name tells us that there is a methyl group coming of the second carbon in a butane molecule as well as the third),  |
** What type of molecule is this?, 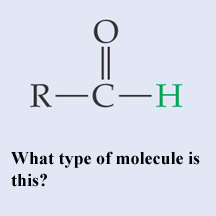 | A basic aldehyde p.48,  |
* What type of molecule is this?,  | carboxylic acid p.48,  |
** What type of molecule is this?, -copy.gif) | aldehyde (hexanal) p.48, -copy.gif) |
**, .gif) | ketone (2-butanone) p.48, .gif) |
** What type of molecule is this?, 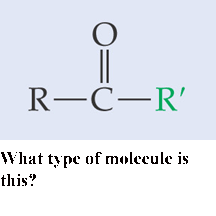 | Ketone p.48, 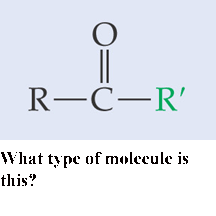 |
| ** C6H6 in a ring structure is _____. | Benzene p.46, 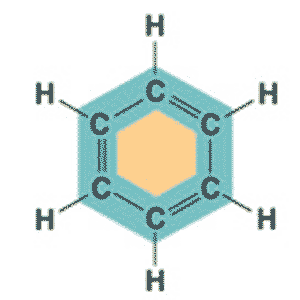 |
| ** What is the molecular formula and shape of benzene? | C6H6, ring p.48 |
| * One of several organic compounds with the same molecular formula but different structures, and therefore, different properties. | isomer p.47 |
| * A functional group present in organic acids consisting of a single carbon atom double-bonded to an oxygen atom and also bonded to a hydroxyl group. | carboxyl group p.48,  |
| ** A functional group consisting of a sulfur atom bonded to a hydrogen atom (--SH). | sulfhydryl group p.48,  |
| * A functional group that is often written as --COOH. | carboxyl group (It's -COOH because it is a C double-bonded to an O and single-bonded to OH) p.48,  |
| * A functional group that consists of a nitrogen atom bonded to two hydrogen atoms. It can act as a base in a solution, accepting a hydrogen ion and acquiring a charge of +1. | amino group p.48, 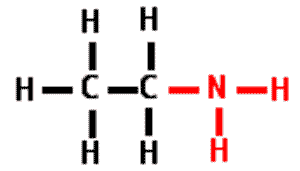 |
| * An organic molecule consisting only of carbon and hydrogen is called a(n) _____. | hydrocarbon (the butane molecule below is a hydrocarbon because it has only hydrogens and carbons) p.46, 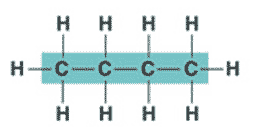 |
| ** An arrangement of two non-carbon atoms, each bound to one of the carbons in a carbon-carbon double bond,where the two non-carbon atoms are on the same side relative to the double bond. | cis p.47, 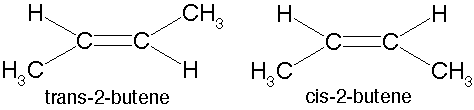 |
| ** An arrangement of two non-carbon atoms, each bound to one of the carbons in a carbon-carbon double bond,where the two non carbon atoms are opposite sides relative to the double bond. | trans p.47, 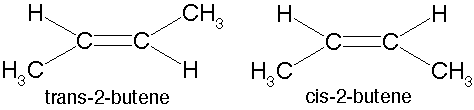 |
| * A specific configuration of atoms commonly attached to the carbon skeleton of organic molecules and usually involved in chemical reactions. | functional group (the carboxyl-group in red below is a functional group that makes the molecule acidic because it often loses the hydrogen as a hydrogen ion into the solution which lowers the pH of the solution) p.49,  |
| * A type of isomer in which carbons have covalent bonds to the same atoms, but these atoms differ in their spatial arrangements due to the inflexibility of carbon-carbon double bonds. | Cis-trans isomers (a.k.a. geometric isomer) p.47, 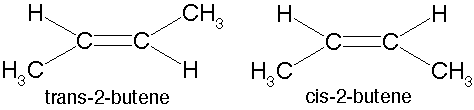 |
| * A functional group that is important in energy transfer. | Phosphate group (think of ATP-->ADP + P + energy) p.48, 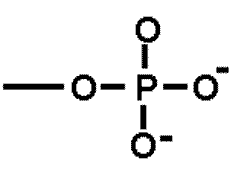 |
| * One of several organic compounds that have the same molecular formula but differ in the covalent arrangement of their atoms. | structural isomer p.47, 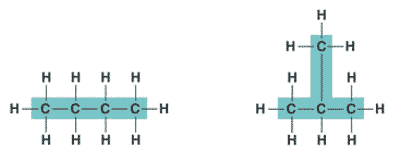 |
| ** A functional group present in aldehydes and ketones that consists of a carbon atom double-bonded to an oxygen atom. | carbonyl group p.48, 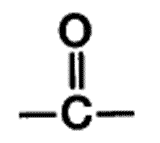 |
| * A functional group consisting of a hydrogen atom joined to an oxygen atom by a polar covalent bond. Molecules possessing this group are soluble in water and are called alcohols. | hydroxyl group p.48,  |
| * One of two molecules that are mirror images of each other. | enantiomer (also called a stereo-isomer) p.48, 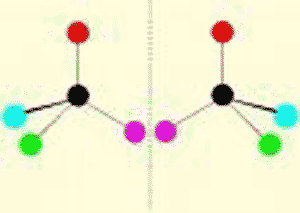 |
| ** A(n) ______ carbon is a carbon atom that is attached to four different atoms or groups of atoms. The result is to allow for the possibility of another molecules that can be an enantiomer (mirror image) of the first molecule. | asymmetric carbon (In the diagram below, the #2 carbon is the asymmetric carbon. Even though it bonds to two carbons, the #1 carbon can be considered an aldehyde GROUP and the number #3 carbon can be considered a propyl group. Therefore, all 4 groups coming off the #2 carbon are different) p.47, 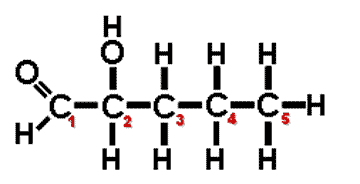 |
** What is the name of the molecule shown below?, 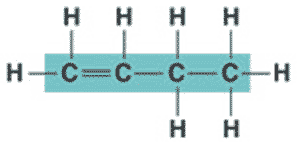 | 1-butene (the 1 signifies that the double bond starts at the first carbon),  |
** What is the name of the molecule shown below?, 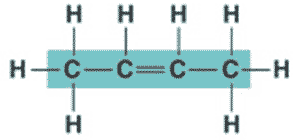 | 2-butene (the 2 signifies that the double bond starts at the second carbon),  |
** What type of molecule is this?,  | An aldehyde (It's an aldehyde because the carbonyl group is coming off a terminal carbon. If it wasn't, it would be a ketone. The name of this molecule is ethanal, since it has two carbons like ethane) p.48,  |
* What is the name of the functional group in red?,  | amino group p.48,  |
** Which carbon on this molecule is the asymmetrical carbon?,  | In the diagram below, the #2 carbon is the asymmetric carbon. Even though it bonds to two carbons, the #1 carbon can be considered an aldehyde GROUP and the number #3 carbon can be considered a propyl group. Therefore, all 4 groups coming off the #2 carbon are different. p.47,  |
** What is the name of this ring structure?, 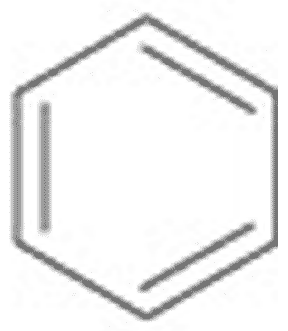 | Benzene (The structure you saw on the other side of this card was an abbreviated structural formula. These formulas assume a carbon at each corner and enough hydrogens to make 4 bonds coming off each carbon. Below is the unabbreviated structural formula for comparison) p.46,  |
** What is the name of this ring structure?,  | Benzene p.46,  |
* What is the name of this hydrocarbon?,  | Butane p.46,  |
** What is the name of this functional group?,  | carbonyl group p.48,  |
** What is the name of this type of molecule and what is the name of its functional group?, 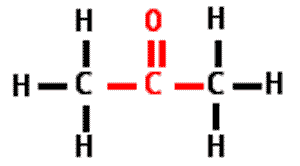 | ketone, carbonyl group (The specific name of the molecule pictured is propanone because it is a ketone with 3 carbons like propane) p.48,  |
* What is the name of this type of molecule and what is the name of its functional group?,  | Carboxylic acid, carboxyl group (This molecule is called ethanoic acid but is more commonly referred to as acetic acid which is the acid in vineger) p.48,  |
** What is this diagram showing?, 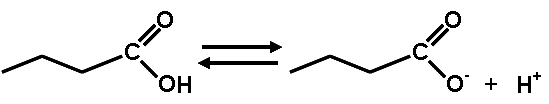 | This shows how carboxylic acids (notice the carboxyl group at the end of the abbreviated structural formula of hydrocarbons) will release a hydrogen ion into water solutions. Hydrogen ions decrease the pH of the solution. That is why it is a carboxylic ACID. p.48 + an activity that will be completed in class, 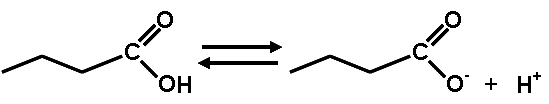 |
What is the name of this molecule?,  | cyclohexane (The structure you saw on the other side of this card was an abbreviated structural formula. These formulas assume a carbon at each corner and enough hydrogens to make 4 bonds coming off each carbon. Below is the unabbreviated structural formula for comparison) p61, 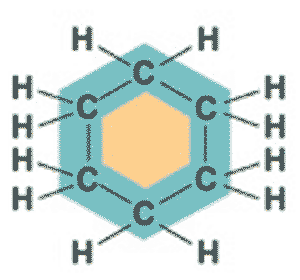 |
** What is the name of this molecule?,  | cyclohexane p.46,  |
* What do we call these two molecules?,  | enantiomers (or stereoisomers; notice that they are mirror images of each other) p.47,  |
* What type of isomers are these?, 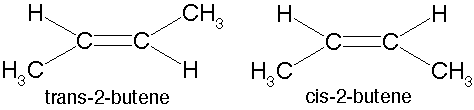 | cis-trans isomers (a.k.a. geometric isomers) p.47, 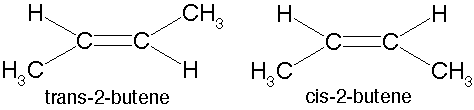 |
** Which of these cis-trans isomers would be considered the "cis" isomer and which would be the "trans" isomer?,  | The one on the left is "cis" and the one on the right is "trans" (the prefix trans means across) p.47,  |
* What is the name of this hydrocarbon?,  | Ethane p.45,  |
* What is the name of this type of molecule and what is the name of its functional group?,  | An alcohol, hydroxyl group (This is ethanol because it has two carbons like ethane. The common name is ethyl alcohol) p.48,  |
* What is the name of this functional group?,  | phosphate group p.48,  |
* What is the name of this hydrocarbon?, 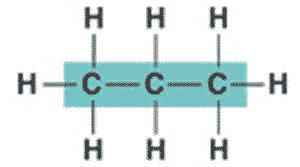 | propane p.46,  |
* What type of isomers are these?,  | structural isomers p.47,  |
** What is the name of this functional group?,  | Sulfhydryl group p.48,  |
| * Carbon can form a wide variety of compounds because it forms ____ covalent bonds. | 4 (p.45) |
* Each line in a structural formula (like the one below) represents a _____., 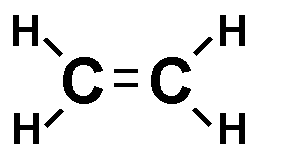 | pair of shared electrons, 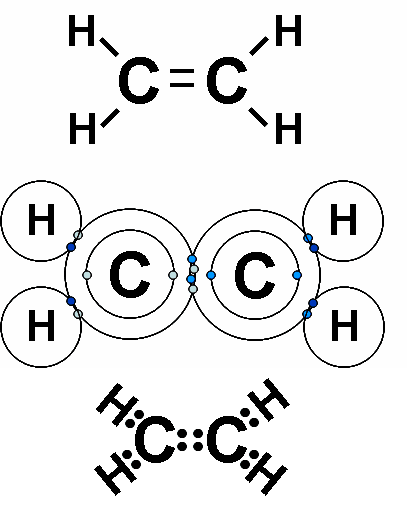 |
| * ____ is the source of carbon for all of the organic molecules found in organisms. | carbon dioxide p.46 |
* Hydrocarbons (like the one shown below) are ______ and _____, therefore they don't dissolve in water.,  | Non-polar, hydrophobic p.46 |
| * An important energy storing molecule that has 3 phosphate groups is called _____. | Adenosine tri-phosphate (ATP) p.49, 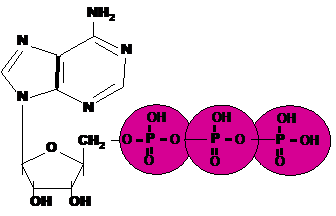 |
| ** Hydrocarbons can be either _____, _____, or _____ shaped. | straight, branched, or ring shaped p.46 |
| * Organic molecules consisting of only carbon and hydrogen are called ______. | hydrocarbons p.46 |
**, 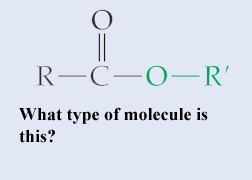 | Ester {The image below is an ester called propyl-ethanoate (you don't need to know how to name these specifically, but you might be able to look at the prefixes involved and figure out the logic). Esters are known for their aromatic qualities (smell) and are used in many perfumes, pheromones, and help give flavor and smell to certain fruits, like apples, bananas, and strawberries. When you take college organic chemistry, you will probably make one of these fruit-smelling ester molecules by mixing an organic acid and an alcohol. Your book doesn't cover this, but I have seen references in AP tests to phosphodiester bonds. These are bonds where a phosphate group helps link one nucleotide to another in DNA or RNA. Knowing what an ester bond is can help you figure out what a phosphodiester bond is}, .gif) |
** What type of bond is shown being formed in the picture below?, 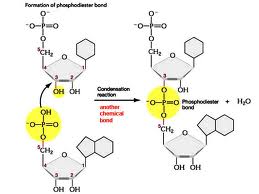 | phosphodiester bond (These are bonds involving a phosphate group (PO4-) that link one nucleotide to another in DNA or RNA. Notice the part of the bond that gives it the ester name. This is not covered in your book, but I have seen it on AP exams, so learn it),  |
| * The repeating building blocks of larger molecules are known as ____. | monomers p.49 |
| * Many monomers linked together make up a ____. | polymer p.49 |
| * A polymer is a large molecule made up of many ____. | monomers p.49 |
| * The root word "poly" means ____. | many |
| * The root word "mono" means ___. | one |
| * The four categories of biological macromolecules are ___, ___, ___, and ___. | carbohydrates, proteins, lipids, and nucleic acids (Although some people, including the authors of this textbook do not consider lipids to be macromolecules) p.44, 
|
| * The type of macromolecule that is used primarily as a source of quick energy is ___. | carbohydrates p.50, 
|
| * Bread, pasta, cereal and fruits are high in which type of macromolecule? | carbohydrates (from your notes), 
|
| * The monomers of complex carbohydrates are ___. | simple sugars (a.k.a. monosaccharides) pp. 51 & 52, 
|
| * Glucose, fructose and galactose are examples of ____. | simple sugars (or monosaccharides - note; these three are all hexose sugars meaning they have six carbons. There are also simple sugars that are pentose sugars with 5 carbons such as ribose and deoxyribose, found in nucleotides. Triose sugars such as glyceraldehyde are important in respiration and photosynthesis) p.51, 
|
| * Sucrose is commonly called ____. | table sugar pp. 51 & 52 |
| * Sucrose is a type of carbohydrate made of two simple sugar monomers (glucose bonded to fructose). Therefore, it is a ___. | disaccharide p.51 |
| * Another term for simple sugars is ___. | monosaccharides p.51, 
|
| * Many monosaccharides bonded together are called a ____. | polysaccharide p.52, 
|
| ** Lactose is a type of disaccharide found in _____. | milk p.52 |
** Plants usually store their carbohydrates as the polysaccharide known as ____., 
| starch p.53 |
** Animals store carbohydrates in their liver and muscles as the polysaccharide known as ___., 
| glycogen p.53, 
|
| * The cell walls of plants are made of the polysaccharide known as ____. | cellulose p.53,  , , 
|
| * _______ is a tough polysaccharide that can only be broken down by bacteria, some protists and some types of fungi. | cellulose pp. 53 & 54,  |
** Cellulose in plant cell walls can only be broken down for energy by ___ ., 
| Bacteria, some protists and certain fungi (Animals that eat grass, which is mostly cellulose, depend on bacteria in their digestive tract to break the cellulose down into sugar monomers that the animals can then digest. Often times, there will be a compartment in their digestive tract that is sectioned off for this purpose) pp73&74,  |
** Glycogen is mainly stored in an animals ___ and ____., 
| muscles and liver p.53 |
* Starch, glycogen and cellulose are all types of _____ and would be classified in the major macromolecule class of _______., 
| Polysaccharides, carbohydrates pp. 52 & 53, 
|
| * Sugars are classified in the macromolecule category called ___. | carbohydrates p.51 & 52, 
|
| * The monomers of proteins are ____. | amino acids p.57, 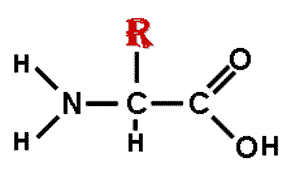 , , 
|
* A long straight chain of amino acids is called a(n) ____., 
| polypeptide (It's not considered a protein until it folds into a specific 3-dimensional shape that allows it to do it's job) p.57, 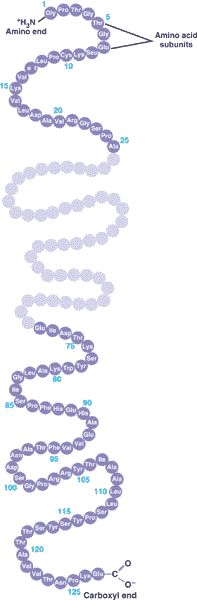 |
* A polypeptide that folds into a 3-D structure that has a specific function is called a(n) ___., 
| protein (The picture below shows the different bonds that hold the protein in its folded shape) p.57, 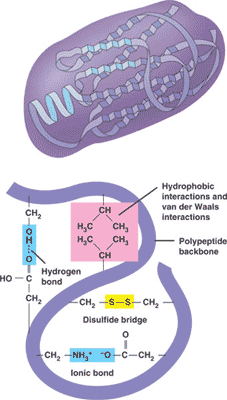 |
** Lean meat is highest in the macromolecule group known as ____., 
| protein (from your notes) |
| * A protein whose shape has been changed due to heat or harsh chemicals is known as a ____ protein. | denatured p.64 |
Organic catalysts (substances that speed up chemical reactions) are known as ___., 
| enzymes p.57, 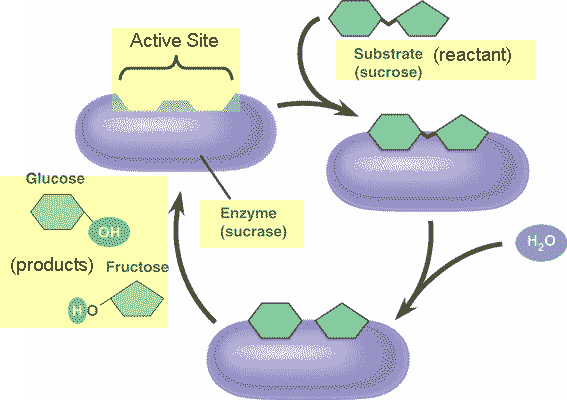 |
| * An enzyme ____ a chemical reaction. | speeds up p.57,  |
| * DNA and RNA are types of ____. | nucleic acids p.66, 
|
| ** The type of molecule that holds an organism's genetic information is called ___ and belongs in the class of macromolecules known as ____. | DNA, nucleic acids p.66,  |
| * The monomers of nucleic acids are ____. | nucleotides p.66,  , , 
|
| ** DNA is made up of four different types of ____. | nucleotides (Adenine, guanine, cytosine, and thymine are the nucleotides in DNA. In RNA, uracil is substituted for thymine. The picture below shows the nitrogenous bases of each which give the nucleotides their names) pp. 66 & 67, 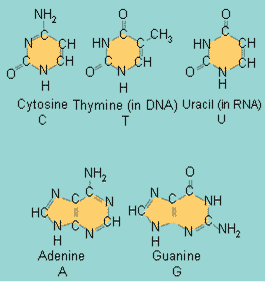 |
| ** Adenine, guanine, cytosine, and thymine are ____ found in ____. | nucleotides, DNA pp. 66 & 67,  |
** The order of nucleotides in a DNA molecule determines the order of ____ in a protein., 
| amino acids pp. 66 & 67,  , , 
|
** A change in the order of nucleotides in DNA is known as a(n) ____., 
| mutation (from your notes) |
** A type of nucleic acid that is usually single-stranded is ___., 
| RNA p.68 |
* A type of nucleic acid that is double stranded is ___., 
| DNA p.68 |
| * Fats, steroids and waxes are classified as ___. | lipids p.54 |
| * The main function of fats in an animals body is to ____. | store energy for later use (other functions can include insulation of the body using fat that lies just under the skin "subcutaneous fat" and cushioning of the organs inside the body cavity) p.56 |
| ** Red meats, dairy and fried foods are high in the category of macromolecules known as ___. | lipids (from your notes) |
| * The building blocks of fats are ___ . | Three fatty acids and one glycerol (The picture below shows a glycerol shaded gray and one long fatty acid chain shaded orange) p.54,  |
| ** Besides being used to store energy, fats are also used for ___. | Insulation and protecting internal organs. (from your notes) |
| ** Fats that are solid at room temperature and are not particularly good for you are a type called ___. | saturated fats p.55,  |
| ** Fats that are liquid at room temperature and are considered healthy to eat are called ___ or _____. | unsaturated fats, oils p.55,  |
| Red meats (like steak) are high in ________ fats. | saturated (p.55 and your notes) |
| ** Fats from vegetable oils and fish tend to be ___. | unsaturated p.55,  |
| ** Phospholipids are major component of the ___. | cell membrane p.56,  |
| * A major component of cell membranes is a type of lipid called a(n) ___. | phospholipid p.56, 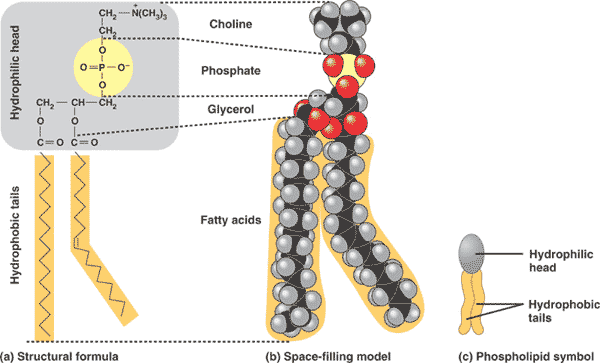 |
** Enzymes are almost always made of ____., 
| proteins p.57 |
** The picture below shows a _______ structure called a(n) ________.,  | secondary structure, alpha helix (keratin, a structural protein in hair and fingernails has alpha helix structure over most of its length) p.62,  |
* The picture below shows the basic structure of a(n) ______.,  | amino acid (notice that your book shows the amino acids in their ionized form as they would be when dissolved in water) p.57,  |
| ** Draw the basic structure of an amino acid and use an R to signify the radical group (where amino acids become different). | p.57,  |
** The picture below shows a _______ structure called a(n) ________., 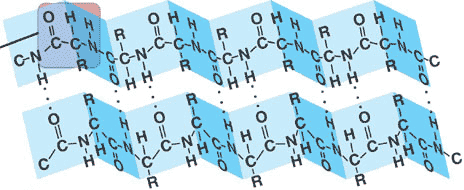 | secondary structure, beta pleated sheet (this structure is found at the core of many globular proteins and in some fibrous proteins such as spider silk) p.62,  |
** The picture below shows the structure of _____., 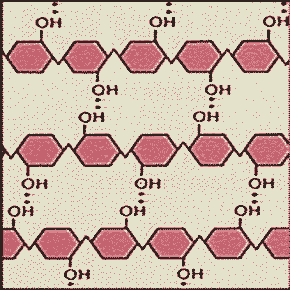 | cellulose (Cellulose is a tough structural carbohydrate which is impossible to digest, unless you are a bacterium, or certain types of protists or fungi, due to the cross-linked bonds that hold the chains together side-by-side) pp.52 & 53,  |
** Some proteins do not assume their final shape until they interact with a specific target molecule. Their flexibility and indefinite structure are important for their function which may require binding with different targets at different times. These proteins are called _______ proteins and are the focus of current research., 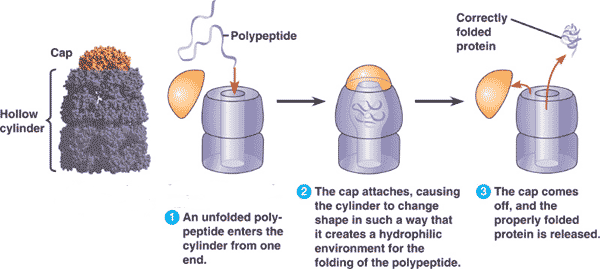 | intrinsically disordered proteins (This is one of the reasons that it can be extremely difficult to synthesize certain protein molecules in the lab. Many drugs are made to fit the shape of a receptor or to attach to the active site of an enzyme in order to change how the enzyme or receptor work, but getting a protein to fold the way it would in the human body can be very difficult without all of the other molecules in the cell that help the protein fold) p.65,  |
** What is the exoskeleton made of that the insect below is trying to molt out of?,  | chitin (Which is a type of structural carbohydrate. It also makes up the exoskeleton of crustaceans like lobster and crab that are closely related to insects. In addition, it is gives the cell walls of fungi their strength and structure, just like cellulose does for plant cell walls) p.54,  |
* The picture is showing a _____ reaction.,  | dehydration reaction (a.k.a. - dehydration synthesis or a condensation reaction) pp. 49 & 50,  |
| ** Two other names for dehydration reaction are _____. | dehydration synthesis and condensation reaction (Synthesis means to put together. In dehydration reactions attach monomers together. There is also water left over, so often times condensation will appear during the reaction if it takes place in a test-tube.) ** This information comes from your notes,  |
* The picture below is showing part of a molecule of _____ which has a ______ shape.,  | DNA, double helix p.68,  |
| ** The monomers of maltose (a disaccharide) are _______. | glucose and glucose (from your notes) |
| ** The monomers of sucrose (a disaccharide) are ___. | glucose and fructose p.52 |
| ** The monomers of lactose are ____. | glucose and galactose (This info can be found in your notes) |
** Glucose and fructose bond to form a _____ called _____., 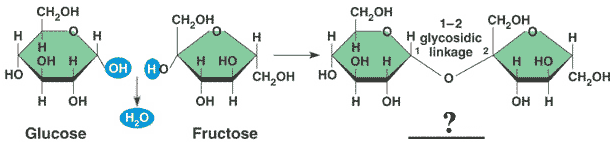 | disaccharide called sucrose (table sugar) p71,  |
** Glucose and glucose bond to form a _____ called _____., 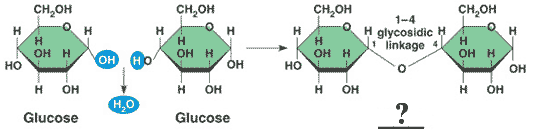 | disaccharide called maltose (malt sugar) * This information is found in your notes,  |
| ** Glucose and galactose bond to form a _______ called _____. | disaccharide called lactose (milk sugar) ** This information can be found in your notes |
** What is the product of this reaction?, 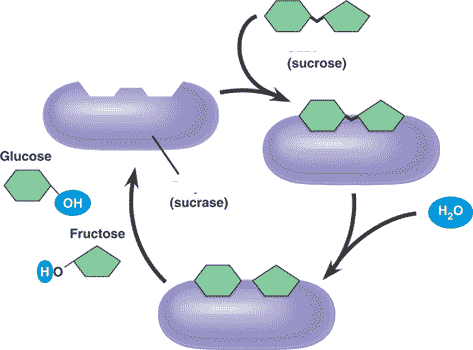 | glucose and fructose (The purple thing is an enzyme that temporarily attaches to the sucrose molecule and helps break the bond that holds the two monomers or sucrose together, thus speeding up the breakdown of sucrose into glucose and fructose),  |
** What is the reactant of this reaction?,  | sucrose (The purple thing is an enzyme that temporarily attaches to the sucrose molecule and helps break the bond that holds the two monomers or sucrose together, thus speeding up the breakdown of sucrose into glucose and fructose),  |
** What is the name of the enzyme in this reaction?,  | sucrase (notice how this enzyme is named after the substrate it breaks down),  |
** Why is water needed for this reaction?,  | Its a hydrolysis reaction (the water is needed to break the bond between the glucose and fructose that make up the sucrose) p.50,  |
| ** Glucose alternates back and forth between a _____ structure and a ______ structure. | straight chain (linear) structure and a ring structure p.51,  |
| ** A triglyceride (or triacylglycerol) is a type of _____ that is made of _____ and _____. | lipid, 1 molecule of glycerol and three fatty acid chains (This picture shows how one glycerol would bond through a condensation reaction to one molecule of fatty acid) pp. 54 & 55,  |
* The reaction shown below is called a(n) _____ reaction.,  | hydrolysis p.50,  |
| * A nucleotide is made of _____. | a phosphate group, a pentose sugar and a nitrogenous base p.66,  |
| ** A nucleoside is made of _______. | A pentose sugar and a nitrogenous base (not the phosphate group) p.66,  |
** What is "A" pointing out?, 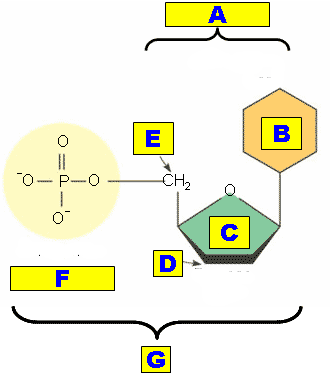 | The nucleoside part of a nucleotide p.66,  |
** What is "B" pointing out?,  | The nitrogenous base part of a nucleotide p.67,  |
** What is "C" pointing out?,  | The pentose sugar part of a nucleotide p.66,  |
** What is "D" pointing out?,  | The 3' (three prime) carbon part of a nucleotide (This is the third carbon of the pentose sugar when you start numbering them from the point of attachment of the nitrogenous base, going clockwise. It is where the phosphate group from another nucleotide would attach using a phosphodiester linkage. The fifth carbon is where this nucleotide's own phosphate group attaches) p.67,  |
** What is "E" pointing out?,  | The 5' (five prime) carbon part of a nucleotide (This is the fifth carbon of the pentose sugar when you start numbering them from the point of attachment of the nitrogenous base, going clockwise. The five prime carbon is where a nucleotide's own phosphate group attaches. The three prime carbon is where the phosphate group from another nucleotide would attach using a phosphodiester linkage) p.67,  |
** What is "F" pointing out?,  | The phosphate part of a nucleotide p.67,  |
* What is "G" pointing out?,  | An entire nucleotide p.67,  |
** What is this and what is it made of?,  | Part of a cell membrane showing a lipid bilayer made of phospholipids p.56,  |
* Where do you find this type of molecule?,  | cell membranes p.56,  |
** This picture shows the _____ structure of a protein.,  | primary structure p.62,  |
** This picture shows the _____ structure of a protein., 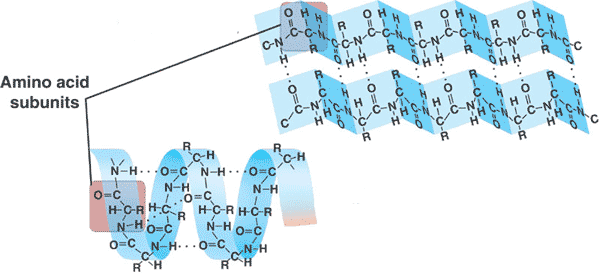 | secondary structure (The one up top is a beta-pleated sheet while the one below is an alpha helix. These are the two different types of secondary structure that proteins can have) p.62,  |
** This picture shows the _____ structure of a protein.,  | tertiary structure p.62 |
** This picture shows the _____ structure of a protein., 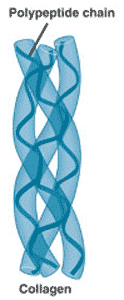 | quaternary structure (notice how the collagen protein, which is a major component of connective tissue, has 3 proteins that form a braid. The alpha-helix structure of the individual proteins give collagen its rubber band-like stretchy characteristics. Old people lose that elasticity in their collagen, leading to wrinkled skin) p.63,  |
** This picture shows the _____ structure of a protein., 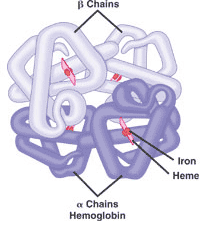 | quaternary structure (This hemoglobin protein is made up of four intertwining polypeptide chains. As the oxygen carrying molecule in red blood cells, hemoglobin also has 4 heme groups with iron at the center to bind directly to oxygen) p.63 |
** "A" is pointing to ____ in this picture showing the process of _____.,  | DNA, protein synthesis (Remember, synthesis means "putting together') p.66,  |
** "B" is pointing to ____ in this picture showing _____.,  | m-RNA (messenger RNA) after it has been transcribed from the DNA template, protein synthesis (Remember, synthesis means "putting together') p.66,  |
** "C" is pointing to ____ in this picture showing _____.,  | m-RNA (messenger RNA) after it has left the nucleus in search of a ribosome, protein synthesis (Remember, synthesis means "putting together') p.66,  |
** "D" is pointing to ____ in this picture showing _____.,  | a ribosome, protein synthesis (Remember, synthesis means "putting together') p.66,  |
** "E" is pointing to ____ in this picture showing _____.,  | a polypeptide chain, protein synthesis (Remember, synthesis means "putting together') p.66,  |
** "F" is pointing to ____ in this picture showing _____.,  | amino acids, protein synthesis (Remember, synthesis means "putting together') p.66,  |
** All of these molecules are _____. The smaller ones in the top row are called _____.,  | nitrogenous bases, pyrimidines (I remember this by thinking that it is easier to measure their "perimeter" since they only have a single ring and perimeter sounds more like pyrimidine that purine) p.67,  |
** All of these molecules are _____. The larger ones in the bottom row are called _____.,  | nitrogenous bases, purines p.67,  |
** The molecule on the top is a(n) ______ while the molecule on the bottom is a(n) ____., 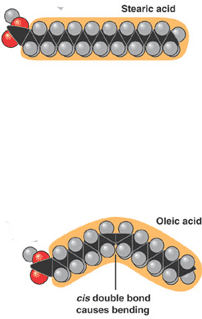 | saturated fatty acid (top), unsaturated fatty acid (bottom) p.55,  |
** Lipids with a ring structure are called _____., 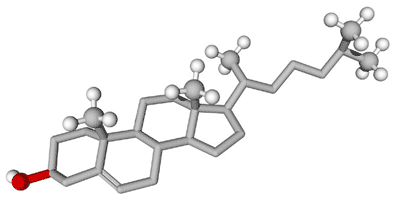 | steroids (cholesterol as well as testosterone and estrogen are steroids) pp. 56 & 57,  |
| ** _____ fats and another type of lipid called _____ may increase the risk of __________ | Saturated fats and cholesterol may increase the risk of atherosclerosis (heart disease) p.57 |
| ** Hydrogenated vegetable oils are _____ fats that have been converted into ____ fats by synthetically adding hydrogens. | unsaturated fats, saturated fats (and also trans fats which may be even worse than saturated fats) pp. 55 & 56 |
* The picture below shows a molecule of _____ which is made of ____ bonded to _____.,  | fat (a.k.a. - a triglyceride), made of glycerol bonded to three fatty acids p.54,  |
| ** Cysteine is an amino acid with a sulfhydryl group as part of its R group. When a protein is folding, two cysteins from different parts of the polypeptide may covalently bond at the sulfhydryl site to form a _______. | disulfide bridge p.63,  |
| * Which macromolecule stores more than twice as much energy per gram compared to the other categories? | Lipids (They have about 9 kilocalories per gram compared with 4 for carbohydrates and proteins) ** see your notes |
| * What is the molecular formula of a hexose sugar such as glucose? | C6H12O6 (fructose and galactose have the same molecular formula as glucose. Therefore, all three are isomers) p.51 |
| ** What is the molecular formula of a disaccharide? | C12H22O11 (When two hexose sugars with molecular formulas of C6H12O6 come together, you have to subtract out 2 hydrogens and one oxygen that are lost as water from the dehydration synthesis reaction) p.52 |
| * The root word "-lysis" or "lyse" means _____ | to break p.50 |
| * To synthesize means to ____. | put together |
| * What trait do all lipids share? | They are hydrophobic (don't mix well with water) p.54 |
| ** Fats that are liquid at room temperature are sometimes called ____ or ______. These types of fats are usually found in ____ and ____. | oils, unsaturated fats, plants and fish. p.55,  |
| ** Fats that are solid at room temperature are made with ______ fatty acids. These types of fats are usually found in ____ other than ____. | saturated, animals, other than fish p.55,  |
| ** Are hydrogenated vegetable oils considered to be saturated or unsaturated fats? | Saturated (When we say an oil has been hydrogenated, what we mean is that the oil has been synthetically converted into a saturated fat by adding hydrogen in the laboratory, and are therefore no better for us than any other saturated fat) pp. 55 & 56 |
| ** In the process of hydrogenating vegetable oils, unsaturated fats are synthetically converted into saturated fats and sometimes a type of unsaturated fat called a ________ that may be even worse for you than saturated fats. | trans fat pp. 55&56 |
| ** A gram of fat stores more than ____ as much energy as a gram of a carbohydrate or protein. | twice (They have about 9 kilocalories per gram compared with 4 for carbohydrates and proteins) p.56 |
| ** In plants, where do you find most of their oils? | in the seeds p.56 |
| ** ______ is a type of steroid that is a crucial component in the structure of cell membranes and can be made in the liver of animals (including you). However, too much in your diet can cause atherosclerosis (hardening of the arteries). | Cholesterol pp. 56 & 57 |
| ** Steroids are a type of _____ made of a carbon skeleton with a(n) ______ structure. | lipid, ring (Some steroids work as hormones which are chemical messengers that travel from one part of the body to another. Most hormones are actually proteins, not lipids like steroids are) pp. 56 & 57,  |
| * Any chemical agent that selectively speeds up chemical reactions is called a(n) _____. | catalyst (Some catalysts are inorganic, like manganese dioxide which can break down hydrogen peroxide, or the metals inside the catalytic converter that convert harmful exhaust from your car into less harmful gases) p.57 |
| ** How many different amino acids do cells use in order to build their proteins? | 20 p.57 |
| ** Bonds between one amino acid joining it to another amino acid are called _____ bonds. | peptide p.60 |
| ** Polypeptide chains fold because of mostly ______ bonds that form between _____ in the polypeptide chain. | weak, side chains, a.k.a R-groups) p.63 |
| ** Disulfide bridges in the tertiary structure of a protein involve covalent bonds between ______ atoms from two cysteine amino acid side chains. | sulfur p.63,  |
| ** The secondary structure of proteins is caused by _____ forming between the ________ of different amino acids in a polypeptide chain. | hydrogen bonds, polypeptide backbones (not the side chains, a.k.a. -R groups) p.62 |
| * When a protein unravels and loses its functional shape due to changes in the environment like high temperatures or pH extremes, a change called _______ has occurred. | denaturation p.64 |
| ** A condition called _______ involving blood hemoglobin is an example of how the shape of a protein can completely change if even just one amino acid in the primary structure of a protein is changed. | sickle cell disease p.64 |
| * At the chemical level, why can a high fever be fatal? | The high temperatures can denature (cause a shape change) proteins in the blood. p.65 |
| ** A nucleic acid polymer can also be called a(n) ____. | polynucleotide p.66 |
| ** The analysis of large sets of genes, or even comparing whole genomes of different species, is an approach called ______. | genomics p.69 |
| ** The analysis of large sets of proteins, including their amino acid sequences, is an approach called _____., | proteomics p.69 |
| ** It would be expected that two species that are evolutionarily closely related (have a recent common ancestor) would have a greater proportion of their ____ and ______ sequences in common that would two less closely related species. | DNA and protein (This is due to the fact that less closely related species have had a longer time since separation for mutations to change their DNA sequences, and therefore their protein sequences) p.69 |