| A | B |
|---|
 | Single Replacement Reaction |
 | Synthesis Reaction |
 | Combination Reaction |
 | Composition Reaction |
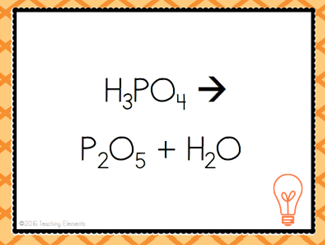 | Decomposition |
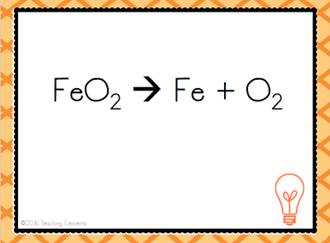 | Decomposition Reaction |
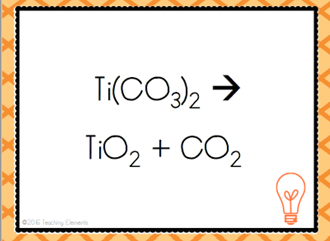 | Decomposition Reaction |
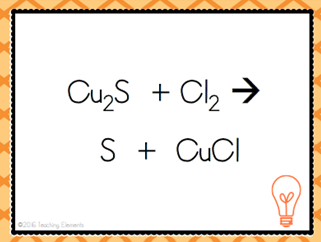 | Single Displacement Reaction |
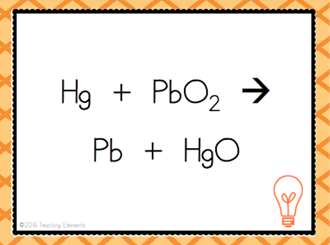 | Single Replacement Reaction |
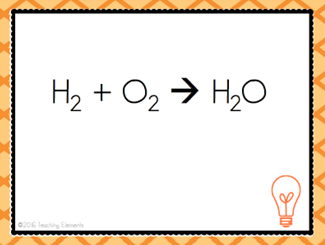
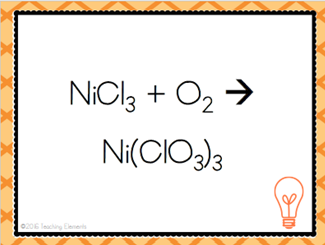
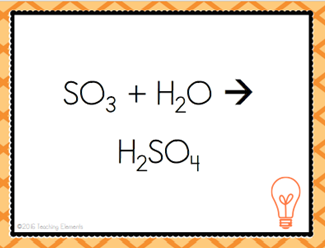
| Two or more substances are combined to form a more complex substance | Synthesis Reaction |
| A + B --> AB | Combination Reaction |
| Opposite of synthesis reaction. A more complex substance breaks apart into simpler ones | Decomposition Reaction |
| AB --> A + B | Decomposition Reaction |
| Usually exothermic (release energy) | Composition Reaction |
| Usually endothermic (absorbs energy) | Decomposition Reaction |
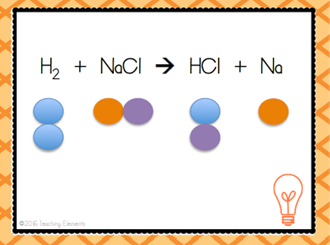
| A + BC --> AC + B | Single Replacement Reaction |
| When a pure substance switches places with another element in a compound | Single Displacement Reaction |
| Fe + S --> FeS | Synthesis Reaction |
| Water decomposes to form hydrogen gas and oxygen gas | Decomposition Reaction |
| Magnesium reacts with water to form hydrogen gas and magnesium hydroxide | Single Replacement Reaction |
| A process in which one or more substances, the reactants, undergo chemical transformation to form one or more different substances, the products | Chemical Reaction |
| A material that is composed of only one type of particle | Pure Substance |
| A chemical substance made up of two or more elements bonded together | Compound |
| A group of two or more atoms covalently bonded together | Molecule |
| A material made up of at least two different pure substances | Mixture |
| Changes involves the change of state | Physical Reaction |
| Anything that occupies space, has mass, and is made up of particles | Matter |