| A | B |
|---|
| Period | Row |
| Group | Family/Column |
| Atomic Radius | Increases down the group & across the period from right to left |
| Ionic Radius | Increases down the group & across the period from left to right |
| Metallic Character | Cu<Ag<Au |
| Nonmetallic Character | I<Br<Cl |
| Electronegativity | Ability of an atom to attract electrons from another atom |
| Electron Affinity | The change in energy when an electron is added to an atom |
| Ionization Energy | Ability of an atom to gain electrons from another atom |
| Cation | Positively charged atom (Metals lose electrons) |
| Anion | Negatively charged atom (Nonmetals gains electrons) |
| Metals | Increased atomic radius and melting/boiling points but decreased ionic radius, electronegativity, electron affinity, and ionization energy |
| Nonmetals | Increased ionic radius, electronegativity, electron affinity, and ionization energy but decreased atomic radius and melting/boiling point |
| Alkali Metals | Group 1 elements with 1 valence electron, form +1 cations, and highly reactive |
| Alkaline Earth Metals | Group 2 elements with 2 valence electrons, form +2 cations, and are reactive |
| Transition Metals | Group 3-12 elements with varying number of valence electrons, form cations, low reactivity, and have high melting/boiling points |
| Halogen | Group 7A elements with 7 valence electrons, form -1 anions, and extremely reactive |
| Noble Gases | Group 8A elements with 8 valence electrons, will not form ions, and are inert/chemically unreactive |
| Lanthanides | 4f elements, corrosive, and heavy metals |
| Actinides | 5f elements, radioactive, heavy metals |
| Aufbau Principle | Begin filling orbitals at ground state (1s) |
| Hund's Rule | Up, up, up...down, down, down |
| Pauli Exclusion Principle | Up, down |
| Standard Notation | Ga: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p1 |
| Noble Gas Notation | Ga: [Ar]4s2 3d10 4p1 |
| Orbital Notation | Picture, 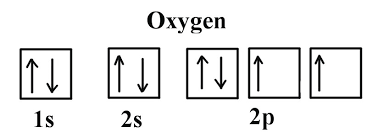 |
| s-orbital | Sphere shaped and holds 2 electrons |
| p-orbital | Dumbbell shaped and holds 6 electrons |
| d-orbital | Clover shaped and holds 10 electrons |
| f-orbital | Flower shaped and holds 14 electrons |
| Atomic Number | Accounts for the number of protons in an atom |
| Atomic Mass Number | Accounts for the number of protons and neutrons in the nucleus of an atom |
| Bohr Model | Picture, 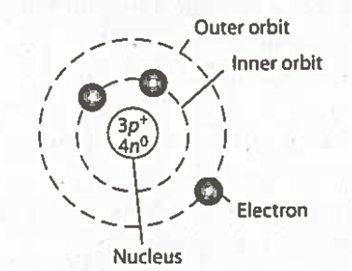 |
| Lewis Dot Diagram | Picture, 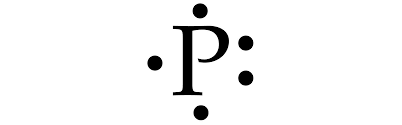 |
| Atom | The simplest unit of matter |
| Compounds | Two or more atoms chemically bonded together |
| Diatomic Molecules | Nonmetals that bond to themselves in order to gain a stable configuration, i.e.BrINClHOF |