WORKSHEET: BOND TRIANGLE - PART II
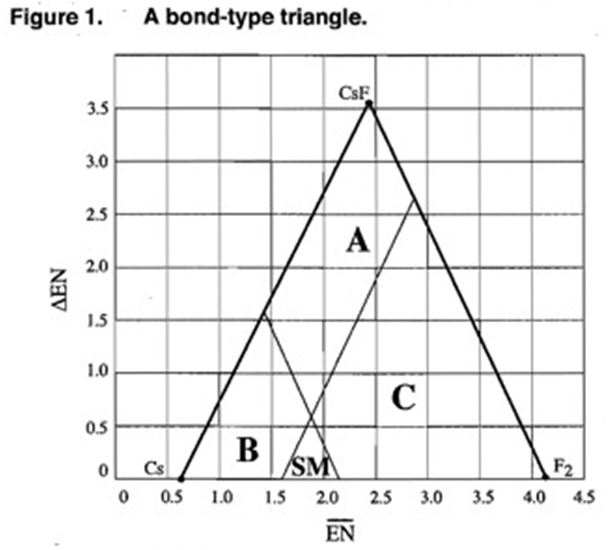
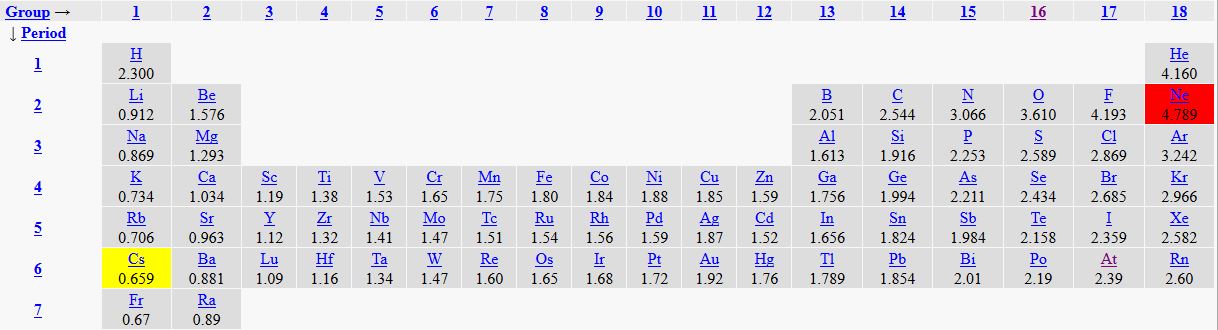
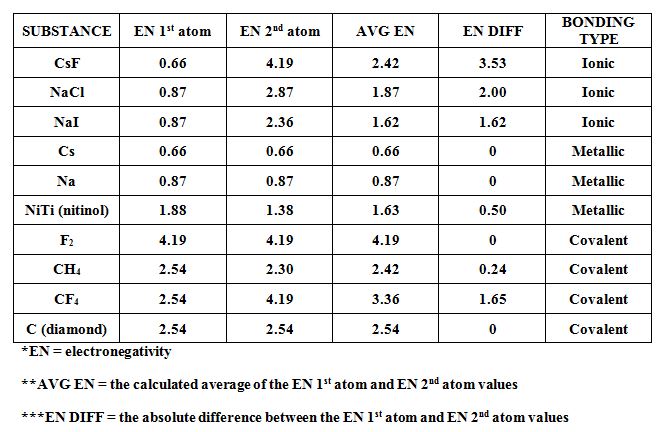
A bond-type triangle is a chart that enables us to predict the properties of a substance based on the electronegativities of the elements that comprise that substance. Since Cs has the lowest electronegativity and fluorine has the highest (not counting neon), the three substances: Cs, CsF and F2 represent extreme cases and are therefore placed at the three extreme corners of this triangle. This triangle can be divided into regions which indicate predominant type of bonding present in substances. The dividing line between these regions are NOT absolute, but they give a general idea of the boundaries between different types of bonding. Many substances have properties that re intermediate between the three bond types: metallic, covalent and ionic. Si (silicon) for example, is know as a semiconductor; it has properties which are intermediate between metallic and covalent.
| Please enter your name. (optional) |
|
|
|